How Does a Storage Battery Work
Introduction
A storage battery, also known as a rechargeable battery, is a type of electrical battery that can be recharged and used multiple times. These batteries are used in a wide range of applications, from powering small electronic devices to storing energy for large-scale renewable energy systems. In this article, we will take a closer look at how storage batteries work.
Chemical Reactions
Electrolytes
Storage batteries contain one or more cells, each of which consists of two electrodes and an electrolyte. The electrodes are typically made of different materials, such as lead and lead dioxide in a lead-acid battery, while the electrolyte is a substance that allows ions to flow between the electrodes.
Charging Process
When a storage battery is being charged, an electric current is applied to the battery, causing a chemical reaction to occur. This reaction converts the materials in the electrodes and electrolyte into a charged state, allowing the battery to store energy.
Discharging Process
Powering Devices
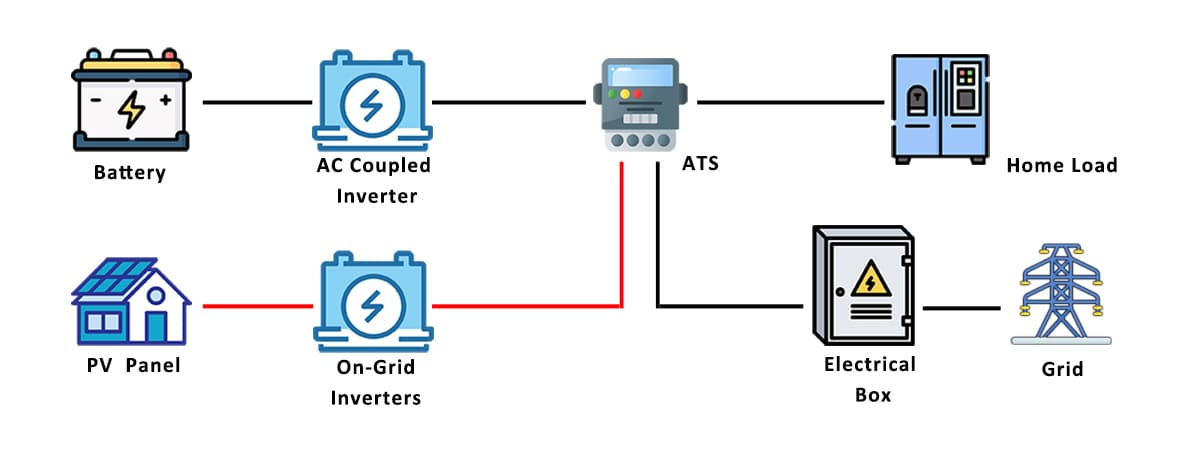
When a storage battery is being used to power a device, the process is reversed. The chemical energy stored in the battery is converted back into electrical energy, which can then be used to power the device.
Recharging
Once a storage battery has been discharged, it can be recharged by applying an electric current to the battery. This reverses the chemical reaction and restores the battery to a charged state, ready to be used again.
Types of Storage Batteries
There are many different types of storage batteries, each with its own unique chemistry and characteristics. Some common types include lead-acid, lithium-ion, nickel-cadmium, and nickel-metal hydride batteries. Each type has its own advantages and disadvantages, making them suitable for different applications.
Conclusion
Storage batteries are an essential part of modern society, providing a reliable and efficient way to store and use electrical energy. By understanding how they work, we can better appreciate the role that storage batteries play in powering our everyday lives and shaping the future of renewable energy.



